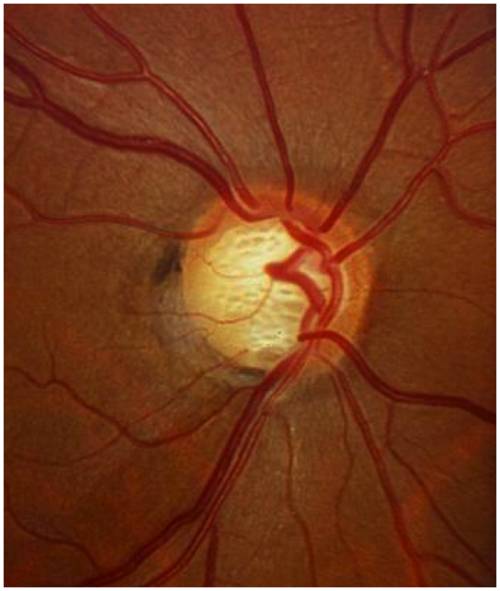
Diagnosis:
Diagnosing glaucoma involves several painless, non-invasive tests:
- Tonometry: Measures the pressure inside your eye.
- Visual Field Test: Checks for vision loss in your peripheral (side) vision.
- Optical Coherence Tomography (OCT): Uses computer imaging to measure the thickness of your optic nerve fibers, detecting damage that can’t be seen with the naked eye.
- Optic Disc Photography: Takes pictures of your optic nerve to monitor any changes over time.
- Pachymetry: Measures the thickness of your cornea, which can affect eye pressure readings.
- Gonioscopy: Examines the drainage angle of your eye to see if it is open or closed.
Treatment:
Medications (First-Line Treatment):
Glaucoma medications aim to lower eye pressure by reducing fluid production or improving fluid drainage in the eye. Common treatments include:
- Prostaglandin Analogues: The most commonly prescribed, these help fluid drain from the eye more effectively.
- Beta-Blockers: Reduce fluid production in the eye.
- Adrenergic Agonists: Increase fluid drainage but are less commonly used due to side effects.
- Cholinergics: Increase fluid drainage but require multiple doses daily and have significant side effects.
- Carbonic Anhydrase Inhibitors: Reduce fluid production, often used if other treatments aren’t effective.
- Combination Products: Medications that combine two different drugs to reduce eye pressure more effectively.
Non-Medication Treatments (Second-Line Treatment):
- Laser Trabeculoplasty: Uses a focused laser to improve drainage and reduce eye pressure, often used if medications aren’t effective.
- Trabeculectomy: Surgery that creates a new drainage path for eye fluid, usually considered if other treatments fail.
- Cyclodestructive Procedures: Used in severe cases, this treatment reduces fluid production by damaging the part of the eye that produces fluid.